Measuring the concentration
Jan Hein HooijschuurLevelBasic
The relation of absorbance to concentration is given by Lambert-Beer's law (Beer's law): A = ɛlc
(where A is absorbance (unitless), ɛ (epsilon) is molar absorption coefficient (or molar absorption constant) of the analyte for a certain wavelength (l⋅mol-1⋅cm-1), l is path length (cm) through your cuvette and c is the concentration of the analyte (mol⋅l-1).)
Demo of how to calculate a concentration from a series of measuments(4:58 mins):
How to generate a standard curve, and use that standard curve to determine the concentration of an unknown solution, titled "Determining the Concentration of an Unknown Sample Using the Standard Curve Excel 2010" (10:58 mins):
Examples of a calculation with Beer's law:
1. A solution of 1 mg paracetamol in 100 ml methanol (MeOH) is measured with UV-Vis.
The intensity of the light through a blank solution of MeOH (I0) is 100% by definition.
The measured intensity of sample solution (I) is 13.18%
Formula: A = - log10T = -log10 (I/I0).
Known values: I0 = 100%
I = 13.18 %
A = -log10 (13.18/100)
A = -log10 (0.1318)
A = -(-0.880)
A = 0.880
2. A solution of 1 mg paracetamol (MW=151.17 g/mol) in 100 ml MeOH (c) is measured in a 10 mm cuvet (l) with UV-Vis.
The absorption maximum of paracetamol in MeOH is at 247nm with a molar absorption coefficient of 12850 l⋅mol-1⋅cm-1 (ɛ).
Formula: A = ɛlc
Known values: ɛ = 12850
l = 10 mm = 1 cm
c = 1 mg/100ml = 10mg/l = 0.01g/l
c = 0.01 × 1/151.17 = 6.61⋅10-5 mol/l
A = 12850 × 1 × 6.61⋅10-5
A = 0.849
Advanced understanding of Beer's law:
For a theoretical explanation on the derivation of the Beer’s law, click this link.
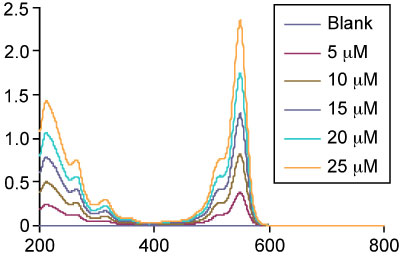
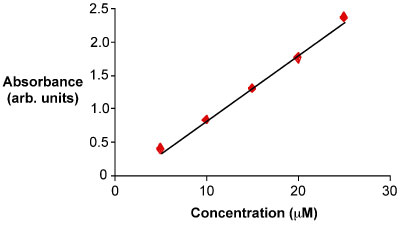
An example of the linear correlation – a straight line - between the concentration of the analyte and the absorbance.
A whiteboard presentation on the calibration curve and how to find an unknown concentration (13:35 mins):
Summary of tips and tricks from the experts We suggest that at this stage you do a virtual UV-Vis measurement. Try this nice java-applet from the website of the chemistry department of the Davidson College in North Carolina, click here[VirtMeas].






