On-line SPE-LC-MS/MS
Rosa MorelloLevelBasic
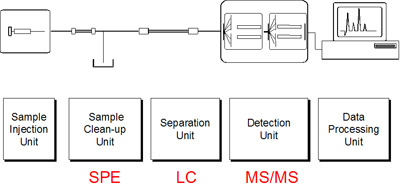
Hyphenation of LC and MS
Why Hyphenation of LC and MS?
- MS is not only an attractive detection method but a powerfull separation technique
- MS relies on a different physical property of the analyte to effect separation, i.e. on the mass-to-charge ratio (m/z) of ions derived from the target analyte(s)
- MS is orthogonal to LC
- Coupling of LC with MS results in a multidimensional separation / detection system
- LC-MS has a very high separation power (peak capacity)
The question is: Can we just dilute and shoot? So, is there any need for sample clean-up ? ! The answer is definitely: yes we need to clean up!
- Fractionate - Switch - Separate
SPE - LC is an attractive combination.
LC-System- Relatively low separation power of conventional analytical columns
Interferences by LMW matrix components - Mobile phases with acidic pH and relatively high amount of organic modifier
- Incompatible with biofluids
- Precipitation or unspecific adsorption of proteins
- Irreversible deterioration of separation column
- MS compatible buffers restrict pH range for separation to pH 3 – 6 (ESI)
MS-Detector
- Ionisation of target analyte(s) can be supressed or enhanced by low molecular weight (LMW) and/or high molecular weight (HMW) matrix components
irreproducible signal(s) - LMW and HMW matrix components not beeing removed or fully separated can cause:
- high baseline noise
- worse signal to noise ratio (reduced sensitivity)
- contamination of MS-source
- Sample or buffer salts not easily volatilised contaminate MS-source causing a loss in sensitivity
Ion Suppression
Understanding and eliminating ion suppression effects is essential for LC-MS analysis of complex biofluids.
In mass spectrometry
- Ion suppression is the result of sample components that diminish the ionization of or compete in the ionization process with the analyte(s) of interest
- Also components of the sample that do not appear in the mass spectrum can cause ion suppression
- In biological fluids, natural variation in endogenous compound concentrations from one sample to another can cause varying levels of ion suppression
- This variation in turn contributes to unacceptable variability in the signal response for the target analyte
Ion suppression in bioanalytical MS
Ion suppression phenomena (“matrix effects”) are directly related to
- unselective sample clean-up and / or
- insufficient / inappropiate chromatographic separation
Monitoring Ion Suppression (Matrix Effects)
Postcolumn Infusion Experiment
1. Infuse a dilute solution of the analyte at a constant rate into the effluent flowing from the LC system to the mass spectrometer.
2. After obtaining a steady baseline, inject a blank sample extract into the LC system Any eluted material that suppresses ionization in the MS will cause a drop in the baseline of the “Infusion Chromatogram”
Sample clean up methods
Infusion Chromatograms with matrix effects: examples of different sample clean-up methods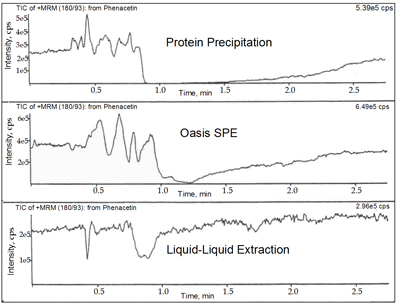 Figure kindly provided by Dr. Richard King, Merck and Co., Inc., Drug Metabolism; West Point, PA, USA Matrix: Human plasma; Analyte: Phenacetin; Mode: ESI-MS/MS
Figure kindly provided by Dr. Richard King, Merck and Co., Inc., Drug Metabolism; West Point, PA, USA Matrix: Human plasma; Analyte: Phenacetin; Mode: ESI-MS/MS
Infusion Chromatograms: Plasma samples originating from different species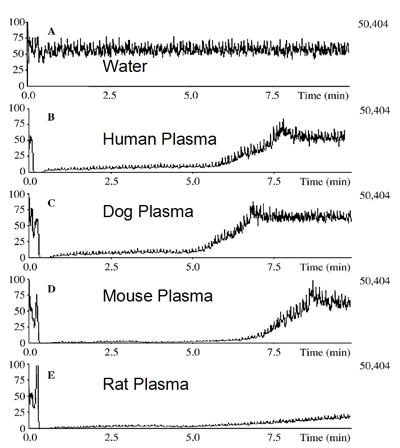 Polson C. et al., J. Chromatogr. B 785 (2003) 263-275Precipitating agent: Acetonitrile; Mobile phase : MeOH / H2O (50/50; v/v); Sample: Plasma/Precipitant (33/66; v/v)
Polson C. et al., J. Chromatogr. B 785 (2003) 263-275Precipitating agent: Acetonitrile; Mobile phase : MeOH / H2O (50/50; v/v); Sample: Plasma/Precipitant (33/66; v/v)
Infusion Chromatograms: Effect of Protein Precipitating Agents on Ion Suppression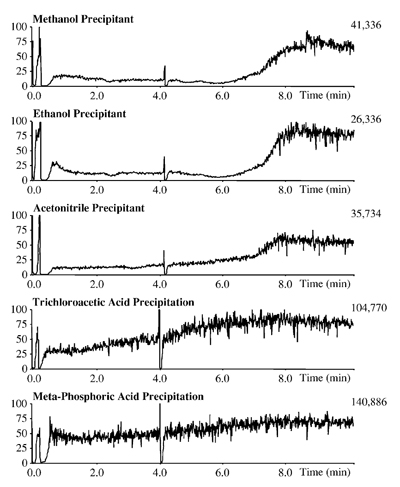 Polson C. et al., J. Chromatogr. B 785 (2003) 263-275Mobile phase: MeOH/H2O (50/50; v/v); Sample: human plasma / precipitant (33/66; v/v)
Polson C. et al., J. Chromatogr. B 785 (2003) 263-275Mobile phase: MeOH/H2O (50/50; v/v); Sample: human plasma / precipitant (33/66; v/v)





