Analysis of biogenic amines in wine
Javier Hernández-Borges, Department of Analytical Chemistry, Nutrition and Food Science, University of La Laguna, SpainAbstract Although high performance liquid chromatography (HPLC) is the major technique used for the determination of BAs in wines, gas chromatography (GC) and capillary electrophoresis (CE) have also been employed in the separation and detection of these compounds in wine. The aim of this chapter is to show/overview the different analytical methods available for their analysis.
Javier Hernández-Borges
Departament of Analytical Chemistry, Nutrition and Food Science, University of La Laguna, 38071, La Laguna, Tenerife, Canary Islands, Spain, jhborges@ull.es
Salvatore Fanali
Istituto di Metodologie Chimiche, Consiglio Nazionale delle Ricerche, Area della Ricerca di Roma, Via Salaria Km 29,300, 00015 Monterotondo Scalo, Rome, Italy, salvatore.fanali@imc.cnr.it
KeywordsBiogenic amines, wine, food safety, food quality
LevelAdvanced
Introduction
Until now, different analytical methods have been developed to determine BAs in foodstuffs and biological samples [10]. Concerning wine analysis, most analytical methods used for the determination of BAs are based on chromatographic methods:
- thin layer chromatography (TLC)
- gas chromatography (GC)
- high-performance liquid chromatography (HPLC, the most used).
- But also capillary electrophoresis (CE)
 methods have been proposed providing good results.
methods have been proposed providing good results.
Regarding their detection, BAs generally show little, if any, ultraviolet (UV) or visible light absorption, fluorescence or electrochemical activity and thus, other methods are required to achieve an appropriate sensitivity in the detection like a derivatization with an appropriate agent or the use of ![]() MS. The large majority of assays employ fluorimetric or UV detection with precolumn or postcolumn derivatization techniques.
MS. The large majority of assays employ fluorimetric or UV detection with precolumn or postcolumn derivatization techniques.
Derivatization
When derivatization is developed, it should be ensured that:
- the derivatizing reaction is complete
- the factors affecting the derivatization (temperature, pH, reaction time, etc) are carefully controlled
- the derivative is stable enough.
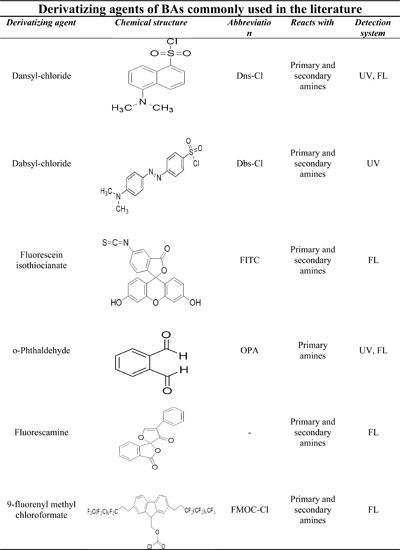
The following table summarizes some of the derivatizing agents most frequently used in these cases (Click for full page view of table).
One of the most used derivatizating agents is DnsCl which reacts with either primary and secondary amino groups, and even tertiary amines in extreme reaction conditions, providing very stable derivatives and combining the unique feature of being both fluorescence and detectable in the UV region. The following figure shows the mechanism of the derivatization reaction:
Derivatization reaction of BAs with dansyl-chloride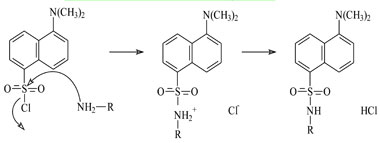
Sample pretreatment
Some sample pretreatment methods in which no derivatization steps or sample cleaning procedures have been proposed in the literature (only dilution and filtration is enough). In this cases either CE as separation technique or MS detection have been used. However, other authors propose different alternatives, especially for the preconcentration or cleaning of the samples.
- Liquid-liquid extraction (LLE): using diethyl ether, ethyl acetate or chloroform (LLE is frequently carried out when GC determination is involved)
- Solid-phase extraction (SPE): using C18 cartridges (especially for HPLC determination)
- Treatment with polyvinylpyrrolidone (PVP): Before sample derivatization and SPE it is frequent to remove phenolic compounds present in the wine (especially red wine) using PVP. For this purpose 0.5 g of PVP is frequently added to 10 mL of wine.
During sample pretreatment and chromatographic/electrophoretic separation also an appropriate internal standard must be chosen, capable of behaviour similar to those of the analytes throughout the whole analytical procedure. In the determination of BAs in wines heptylamine, 1,7-diaminoheptano or cycloheptylamine have been used as internal standards since they are not present in wines.
Determination by HPLC
HPLC technology has been the most popular analytical approach to assay BAs either individually or in combination for the analysis of wines (by reversed-phase-liquid-chromatography, RPLC), also for other sample matrices. A number of different detection systems have been used for this purpose:
- UV and diode array detection
- Fluorescence detection
- Electrochemical detection (used in few occasions)
- Mass spectrometry detection
Most stationary phases are based on silica that has been chemically modified with octadecyl (C18) chains. When conventional C18 columns are used (100-250 cm length, 3.0-4.6 mm i.d), triethylamine (TEA) is frequently used (percentages around 0.25% w/v) as additive of the mobile phase (pH values around 5), in order to avoid the adsorption of these basic compounds onto the stationary phase (caused mainly by the remaining silanol groups) and thus preserve the analytical column lifetime.
RP-HPLC-FL chromatograms of (A) white wine and (B) red wine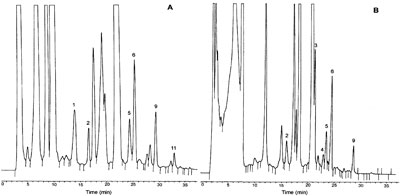 (A) white wine; peaks: 1, methylamine (0.119 mg/L); 2, ethylamine (0.082 mg/L); 5, isoamylamine (0.145 mg/L); 6, putrescine (0.343 mg/L); 9, 1,7-diaminoheptane internal standard (0.843 mg/L); 11, spermidine (0.059 mg/L); and (B) red wine; peaks: 2, ethylamine (0.098 mg/L); 4, 2-phenylethylamine (0.178 mg/L); 5, isoamylamine (0.174 mg/L); 6, putrescine (0.432 mg/L); 9, 1,7-diaminoheptane internal standard (0.843 mg/L); λex=320 nm, λem=523 nm; column, Inertsil ODS-3 (250×4 mm I.D., 5 μm); mobile phase, acetonitrile (40–80% from 0–25 min, 80–100% from 25–30 min, 100–40% from 30–35 min)–water; injection solvent, acetonitrile; injection volume, 20 μl. Reprinted from [11] with permission from Elsevier.
(A) white wine; peaks: 1, methylamine (0.119 mg/L); 2, ethylamine (0.082 mg/L); 5, isoamylamine (0.145 mg/L); 6, putrescine (0.343 mg/L); 9, 1,7-diaminoheptane internal standard (0.843 mg/L); 11, spermidine (0.059 mg/L); and (B) red wine; peaks: 2, ethylamine (0.098 mg/L); 4, 2-phenylethylamine (0.178 mg/L); 5, isoamylamine (0.174 mg/L); 6, putrescine (0.432 mg/L); 9, 1,7-diaminoheptane internal standard (0.843 mg/L); λex=320 nm, λem=523 nm; column, Inertsil ODS-3 (250×4 mm I.D., 5 μm); mobile phase, acetonitrile (40–80% from 0–25 min, 80–100% from 25–30 min, 100–40% from 30–35 min)–water; injection solvent, acetonitrile; injection volume, 20 μl. Reprinted from [11] with permission from Elsevier.
Use of nano-liquid chromatography for the determination of BAs
New miniaturized separation methods offer the possibility to achieve good resolutions and high efficiency in a short analysis time making use of minute volumes of both samples and reagents. This is the case of nano-Liquid Chromatography (nano-LC). In nano-LC capillaries, containing selected stationary phases, i.d. in the range of 10-100 μm and flow rates in the order of nL are currently used. Because the low flow rate hyphenation with specific and very sensitive detectors such as mass spectrometry (MS) can be easily done. Up to now, for BAs nano-LC has only been used in two cases: one of them using UV detection [7] while the other one used MS detection [12].
The following figure shows the nano-LC-UV chromatogram of a group of 10 BAs (ethanolamine, methylamine, tryptamine, 2-phenylethylamine, putrescine, cadaverine, histamine, tyramine, spermidine and spermine). The chromatographic separation of the above reported amines was carried out in a capillary column (100 μm i.d.) packed with bidentate C18 particles. Dns-Cl was also used as derivatizing agent.
Nano-LC chromatogram of a mixture of ten BAs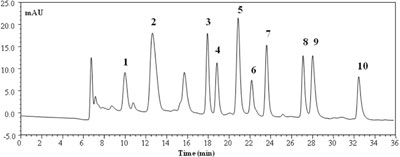 Peak identification: (1) ethanolamine; (2) Methylamine; (3) Tryptamine; (4) 2-phenylethylamine; (5) Putrescine; (6) Cadaverine; (7) Histamine; (8) Tyramine; (9) Spermidine; (10) Spermine. Concentration: aprox. 2 mg/L of each amine except for peak 4 (1.1 mg/L), peak 6 (0.8 mg/L), peak 7 (2.8 mg/L), peak 8 (1.2 mg/L) and peak 9 (3.8 mg/L). UV detection at 250 nm. Redrawn from [7] with permission from Elsevier.
Peak identification: (1) ethanolamine; (2) Methylamine; (3) Tryptamine; (4) 2-phenylethylamine; (5) Putrescine; (6) Cadaverine; (7) Histamine; (8) Tyramine; (9) Spermidine; (10) Spermine. Concentration: aprox. 2 mg/L of each amine except for peak 4 (1.1 mg/L), peak 6 (0.8 mg/L), peak 7 (2.8 mg/L), peak 8 (1.2 mg/L) and peak 9 (3.8 mg/L). UV detection at 250 nm. Redrawn from [7] with permission from Elsevier.
Schematic illustration of the nano-LC system used in [7].![Schematic illustration of the nano-LC system used in [7].](/dchro/gfx/ZamxiayHkC.jpeg)
Determination by GC
GC has also been used for the analysis of volatile amines and their derivatives (i.e. putrescine, cadaverine, histamine, tyramine, spermidine etc…) in wine but not as often as HPLC. The detection systems that have been used for this purpose are basically:
- Flame ionization detection (FID)
- Mass spectrometry detection
Although historically amines present difficulties in GC analysis, it is possible to detect them if they are previously ![]() derivatized. A number of methods can be employed to derivatize amines such as:
derivatized. A number of methods can be employed to derivatize amines such as:
- acylation (with either an acid anhydride, acyl chloride, acyl imidazole, or an acyl amide)
- silylation (with either N,O-bis(trimethylsilyl)trifluoroacetamide (BSTFA) or N-methyl-N-(tert-butyldimethylsilyl)trifluoroacetamide (MTBSTFA))
- carbamate formation (with alkyl chlorformate reagents).
Determination by capillary electromigration methods
CE is also an attractive alternative for BAs determination because of its inherent advantages:
- low sample and reagent requirements
- high speed of analysis
- high efficiency
- high resolving power.
However, the LODs achieved are higher when compared to HPLC or GC, due to the lower amounts of sample injected and to the short optical path length. That drawback can be avoided employing a suitable preconcentration method or using a highly sensitive detection systems.
Because BAs are weak bases, pH values close to 4 are appropriate to protonate the compounds and thus their separation by capillary zone electrophoresis (CZE) is feasible. In this case separation occurs as solutes migrate at different velocities through the capillary due to the difference in their charge to mass ratio. Suitable buffers may contain either phosphate (pKa1 2.1, pKa2 7.2, pKa3 12.3), formate (pKa 3.8), acetate (pKa 4.8), citrate (pKa1 3.1, pKa2 4.7) etc. However, when derivatized amines are analyzed, the whole structure of the derivatized compound should be taken into account since a variation of the pKa value or even a change in the charge may occur. In general, CZE parameters that should be optimized are: the ionic strength of the BGE, its pH value, the presence of organic modifiers like ethanol, methanol, propanol, etc., as well as the separation voltage, temperature, injection time and type (electrokinetic or hydrodynamic) and the washing protocol at the beginning and at the end of the day and between injections.
Up to now, only few works have shown the determination of BAs in wines by CE (the number is much lower than the ones dealing with HPLC). Basically CZE [8, 15, 16] and micellar electrokinetic chromatography (MEKC) [17, 18] modes have been proposed.
The detection systems that have been used for this purpose are basically:
- UV and diode array detection
- Laser induced fluorescence detection (LIF)
- Electrochemical detection
- Mass spectrometry detection (only two works [8, 19]).
CE–TOF-MS extracted ion electropherograms of five standard BAs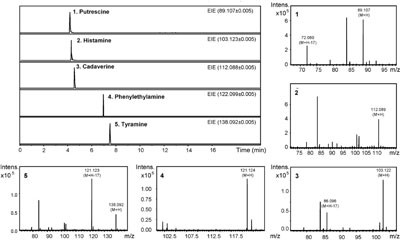 Concentrations: putrescine (10 µg/mL), histamine (1 µg/mL), cadaverine (10 µg/mL), phenylethylamine (0.4 µg/mL), tyramine (1 µg/mL). Separation was performed at +28 kV in a BGE with next composition: 1M formic acid at pH 2 (adjusted with ammonium hydroxide). Reprinted from [8] with permission from Elsevier.
Concentrations: putrescine (10 µg/mL), histamine (1 µg/mL), cadaverine (10 µg/mL), phenylethylamine (0.4 µg/mL), tyramine (1 µg/mL). Separation was performed at +28 kV in a BGE with next composition: 1M formic acid at pH 2 (adjusted with ammonium hydroxide). Reprinted from [8] with permission from Elsevier.
Some examples of methods
Some examples of different methods. Click figure to enlarge.
References
[1] ten Brink, B.; Damink, C.; Joosten, H.M.L.J.; Huis in’t Vent, J.H.; “Occurrence and formation of biologically active amines in foods”. International Journal of Food Microbiology, 1990, 11, 73-84.
[2] Hornero-Méndez, D.; Garrido-Fernández, A.; “Biogenic amines in table olives. Analysis by high-performance liquid chromatography”. The Analyst, 1995, 119, 2037-2041.
[3] McCloskey, D.E.; Woster, P.M.; Casero, R.A.J.; Davidson, N.E.; “Effects of the Polyamine Analogues N1 -Ethyl-N11-((cyclopropyl)methyl)-4,8-diazaundecane and N1-Ethyl-N11-((cycloheptyl)methyl)-4,8- diazaundecane in Human Prostate Cancer Cells”. Clinical Cancer Research 2000, 6, 17-23.
[4] Nairn, L.M.; Lindsay, G.S.; Woster, P.M.; Wallace, H.M.; “Cytotoxicity of novel unsymmetrically substituted inhibitors of polyamine biosynthesis in human cancer cells”. Journal of Cellular Physiology, 2000, 182, 209-213.
[5] Tarantola, G.; “Separazione ed identificazione cromatografica degli aminoacidi negli vini”. Accademia Italiana della Vite e del Vino 1954, 6, 146-157.
[6] Soufleros, E.H.; Bouloumpasi, E.; Zotou, A.; Loukou, Z.; “Determination of biogenic amines in Greek wines by HPLC and ultraviolet detection after dansylation and examination of factors affecting their presence and concentration”. Food Chemistry, 2007, 101, 704-716.
[7] Hernández-Borges, J.; D’Orazio, G.; Aturki, Z.; Fanali, S.; “Nano-liquid chromatography analysis of dansylated biogenic amines in wines”. Journal of Chromatography A, 2007, 1147, 192-199.
[8] Simó, C.; Moreno Arribas, M.V.; Cifuentes, A.; “Ion-trap versus time-of-flight mass spectrometry coupled to capillary electrophoresis to analyze biogenic amines in wine”. Journal of Chromatography A, 2008, 1195, 150-156.
[9] Fernandes, J.O.; Ferreira, M.A.; “Combined ion-pair extraction and gas chromatography–mass spectrometry for the simultaneous determination of diamines, polyamines and aromatic amines in Port wine and grape juice”. Journal of Chromatography A, 2000, 886, 183-195.
[10] Oguri, S.; “Electromigration methods for amino acids, biogenic amines and aromatic amines”. Journal of Chromatography B, 2000, 747, 1-19.
[11] Loukou, Z.; Zotou, A.; “Determination of biogenic amines as dansyl derivatives in alcoholic beverages by high-performance liquid chromatography with fluorimetric detection and characterization of the dansylated amines by liquid chromatography–atmospheric pressure chemical ionization mass spectrometry”. Journal of Chromatography A, 2003, 996, 103-113.
[12] Song, Y.; Quan, Z.; Liu, Y.M.; “Assay of histamine by nano-liquid chromatography/tandem mass spectrometry with a packed nanoelectrospray emitter”. Rapid Communication in Mass Spectrometry, 2004, 18, 2818-2822.
[13] Fernandes, J.O.; Ferreira, M.A.; “Determination of histamine in part wines and grape juices by ion-pair extraction and stable isotope dilution GC-MS”. Chromatographia, 2000, 52, 77-81.
[14] Paik, M.J.; Choi, Y.; Kim, K.R.; “Simultaneous profiling analysis of alkylphenols and amines by gas chromatography and gas chromatography–mass spectrometry”. Analytica Chimica Acta, 2006, 560, 218-226.
[15] García-Villar, N.; Saurina, J.; Hernández-Cassou, S.; “Capillary electrophoresis determination of biogenic amines by field-amplified sample stacking and in-capillary derivatization”. Electrophoresis, 27, 2006, 474-483.
[16] Male, K.B.; Luong, J.H.T.; “Derivatization, stabilization and detection of biogenic amines by cyclodextrin-modified capillary electrophoresis–laser-induced fluorescence detection”. Journal of Chromatography A, 2001, 926, 309-317.
[17] Zhang, N.; Wang, H.; Zhang, Z.X.; Deng, Y.H.; Zhang, H.S.; “Sensitive determination of biogenic amines by capillary electrophoresis with a new fluorogenic reagent 3-(4-fluorobenzoyl)-2-quinolinecarboxaldehyde”. Talanta, 2008, 76, 791-797.
[18] Kovacs, A.; Simon-Sarkadi, L.; Ganzler, K.; “Determination of biogenic amines by capillary electrophoresis”. Journal of Chromatography A, 1999, 836, 305-313.
[19] Santos, B.; Simonet, B.M.; Ríos, A.; Valcárcel, M.; “Direct automatic determination of biogenic amines in wine by flow injection-capillary electrophoresis-mass spectrometry”. Electrophoresis, 2004, 25, 3427-3433.
[20] Pereira, V.; Pontes, M.; Cámara, J.S.; Marques, J.C.; “Simultaneous analysis of free amino acids and biogenic amines in honey and wine samples using in loop orthophthalaldeyde derivatization procedure”. Journal of Chromatography A, 2008, 1189, 435-443.